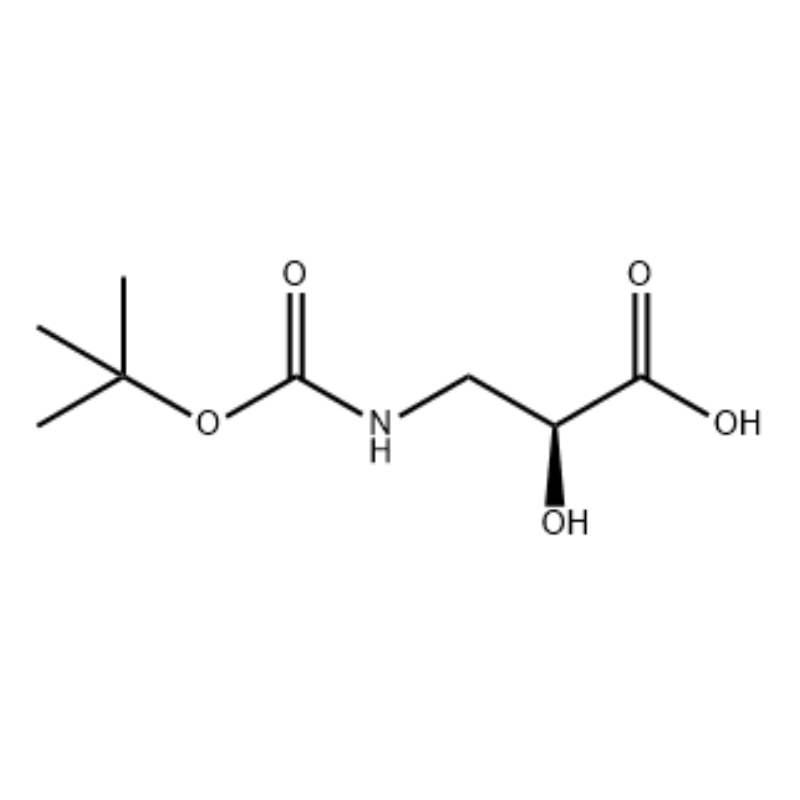
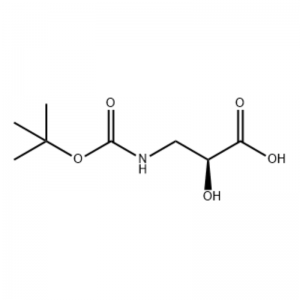
(S)-isoserine 15a (21 g, 0.20 mol) was dissolved in tetrahydrofuran (100 mL)And a mixed solvent of 10percent aqueous sodium hydroxide solution (100 mL),Di-tert-butyl dicarbonate (50 mL, 0.22 mol) was added dropwise.The reaction was carried out at room temperature for 9 hours.The aqueous phase was adjusted to pH 2 with 4 mol/L hydrochloric acid, and extracted with dichloromethane/methanol (v/v = 5/1, 50 mL × 3) and dried over anhydrous sodium sulfate.Filter by suction, concentrate under reduced pressure,The title compound 15b was obtained as a colorless oil (35 g, yield: 85percent).
To a stirring solution of S-isoserine (4.0 g, 0.038 mol) in dioxane: H2O (100 mL, 1:1 v/v) at 0° C was added N-methylmorpholine (4.77 mL, 0.043 mol), followed by BoC2O (11.28 mL, 0.049 mol) and the reaction was stirred overnight with gradual warming to room temperature. Glycine (1.0 g, 0.013 mol) was then added and the reaction was stirred for 20 min. The reaction was cooled to O0C and sat aq. NaHCO3 (75 mL) was added. The aqueous layer was washed with ethyl acetate (2 x 60 mL) and then acidified to pH 1 with NaHSO4. This solution was then extracted with ethyl acetate (3 x 70 mL) and these combined organic layers were dried over Na2SO4, filtered and concentrated to dryness to give the desired N-Boc-3-ammo-2(S)-hydroxy- propanoic acid (6.30 g, 0.031 mmol, 81.5 percent yield): 1H NMR (400 MHz, CDC13) δ 7.45 (bs, 1 H), 5.28 (bs, 1 H), 4.26 (m, 1 H), 3.40-3.62 (m, 2 H), 2.09 (s, 1 H), 1.42 (s, 9 H); 13C NMR (IOO MHz, CDC13) δ 174.72, 158.17, 82, 71.85, 44.28, 28.45.
N-Boc-3-amino-2(S)-hydroxy-propionic acid ; <n="62"/>To a stirring solution of S-isoserine (4.0 g, 0.038 mol) in dioxane: H2O (100 mL, 1 :1 v/v) at 0° C was added N-methylmorpholine (4.77 mL, 0.043 mol), followed by BoC2O (11.28 mL, 0.049 mol) and the reaction was stirred overnight with gradual warming to room temperature. Glycine (1.0 g, 0.013 mol) was then added and the reaction was stirred for 20 min. The reaction was cooled to 0°C and sat aq. NaHCO3 (75 mL) was added. The aqueous layer was washed with ethyl acetate (2 x 60 mL) and then acidified to pH 1 with NaHSO4. This solution was then extracted with ethyl acetate (3 x 70 mL) and these combined organic layers were dried over Na2SO4, filtered and concentrated to dryness to give the desired N-Boc-3-amino-2(5)-hydroxy- propanoic acid (6.30 g, 0.031 mmol, 81.5 percent yield): 1H NMR (400 MHz, CDC13) δ 7.45 (bs, 1 H), 5.28 (bs, 1 H), 4.26 (m, 1 H), 3.40-3.62 (m, 2 H), 2.09 (s, 1 H), 1.42 (s, 9 H); 13C NMR (100 MHz, CDC13) δ 174.72, 158.17, 82, 71.85, 44.28, 28.45.
 Building 12, No.309, South 2nd Road, Economic Development Zone, Longquanyi District, Chengdu, Sichuan, China.
Building 12, No.309, South 2nd Road, Economic Development Zone, Longquanyi District, Chengdu, Sichuan, China. amy@enlaibio.com / cynthia@enlaibio.com / edison@enlaibio.com / daisy@enlaibio.com
amy@enlaibio.com / cynthia@enlaibio.com / edison@enlaibio.com / daisy@enlaibio.com +86 (028) 84841969
+86 (028) 84841969 +86 135 5885 5404
+86 135 5885 5404




















.png)


